  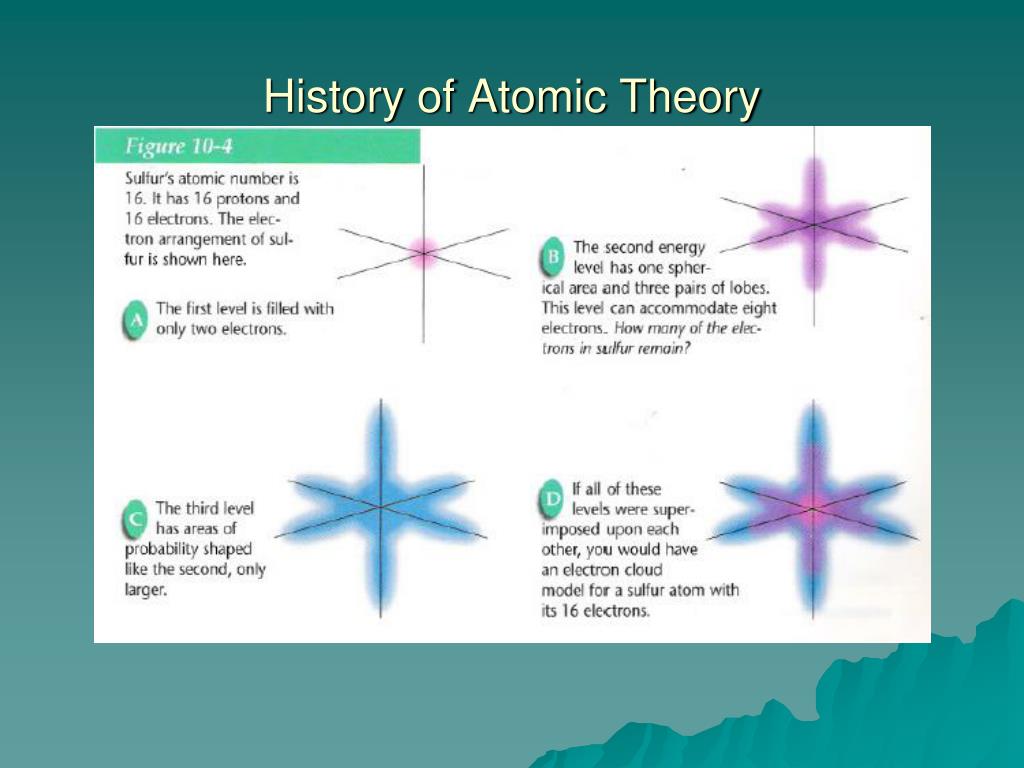
He named the positively charged particles in the atom as protons, and in 1932, another scientist, James Chadwick found a third neutral particle called neutrons. This is because the mass of the nucleus is relatively large, so the particles which come directly towards it will go in a reverse path. However, if the particle approaches the dense and positive core, which is the nucleus in the atom, it will gain a repulsion force and bounce back at a bigger angle. Rutherford then explained that maybe most of the atom is empty so that most of the particles pass the gold with little deflection. 
SOLID SPHERE MODEL PLUM PUDDING MODEL NUCLEAR MODEL PLANETARY MODEL QUANTUM MODEL JOHN DALTON J.J. Was the first, with the help of his teacher Leucippus, to propose an atomic universe. A HISTORY OF THE ATOM: THEORIES AND MODELS How have our ideas about atoms changed over the years This graphic looks at atomic models and how they developed. 460-c.370 BCE) He was a Greek philosopher and younger contemporary of Socrates. The properties of the atoms are determined in direct relationship with the phenomena to be explained. many theories and discoveries have been made about atoms throughout the years. In the history of atomism the 17th century occupies a special place for two reasons: it saw the revival of Democritean atomism, and it saw the beginning of a scientific atomic theory. All attention is focused on the explanation of concrete phenomena. Atomism - Modern Science, Pioneering Work: Modern atomism arose with the flowering of science in the present sense of the word. This was contradictory to Thomson’s model because the particles should have passed the gold atom with very little deflection. Atomism - Modern Theory, Particles, Structure: With the development of a scientific atomic theory, the general philosophical problems gradually disappeared into the background. He observed that most of the alpha particles had little or no deflection, but there were a few particles deflected at a large scale. In 1910, Rutherford shot a thin layer of gold with alpha particles (a kind of positively charged particle) and surrounded the gold with a detecting screen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
